Saving Severely Injured Limbs in Military Settings
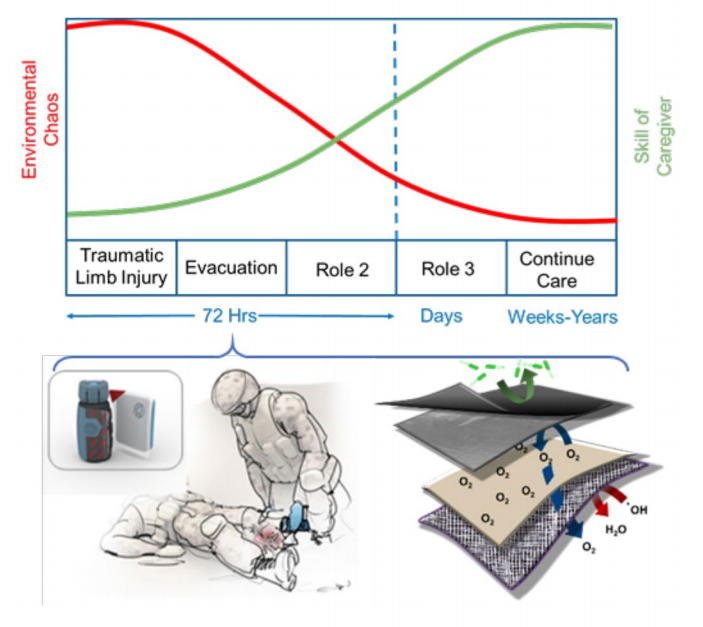 Figure 1. The graph illustrates the inverse relationship between the environmental chaos and the sophistication of treatment over the ensuing time course after a serious limb injury. This results in the need for a lightweight, easily deployable wound dressing that can preserve a severely injured limb in the first 72 h. The dressing can be used with a tourniquet and serves to stop bleeding, prevent infection, provide oxygen, and scavenge radical oxygen species.
Figure 1. The graph illustrates the inverse relationship between the environmental chaos and the sophistication of treatment over the ensuing time course after a serious limb injury. This results in the need for a lightweight, easily deployable wound dressing that can preserve a severely injured limb in the first 72 h. The dressing can be used with a tourniquet and serves to stop bleeding, prevent infection, provide oxygen, and scavenge radical oxygen species.Traumatic limb injuries often occur in chaotic battlefield environments in which a soldier may not be transported to definitive care centers for up to 72 h (Fig. 1). The Acute Care Cover for Severely Injured Limbs (ACCSIL) is intended to fill the gap of medical care during this time span, providing medics with sophisticated tools in a lightweight, easily deployable system able to stop bleeding, prevent infection, regulate oxygen, and ultimately preserve viable tissue until the soldier is transported to a hospital. The ACCSIL system comprises an integrated device offering several benefits to the soldier. The outer cover provides bacterial resistance to prevent infection. The next layer contains an oxygen-generating polymer hydrogel component that can be triggered to release oxygen at a steady rate over 72 h upon application to a wound bed. The final layer is a bioactive gauze that is infused with compounds that can reduce oxidative stress by free radical scavenging while providing further antimicrobial functionality.
Role of Oxygen in Saving a Limb
Oxygen regulation is one of the key functions of this dressing. Especially when a tourniquet is used and the tissue beneath the tourniquet is wrapped in gauze and other protective material, lack of oxygen (hypoxia) can lead to tissue death.1,2 On the other hand, the presence of reactive oxygen species (ROS) can also lead to tissue death through oxidative stress.3 For ACCSIL, scientists have developed a polymer hydrogel that can store latent oxygen in a compact, lightweight polymer and deliver it to a wound bed on demand. At the same time, they have developed a complementary bioactive gauze capable of scavenging ROS. There are natural antioxidant enzymes the body uses to scavenge ROS; Battelle’s device uses a much more stable and robust inorganic antioxidant to functionalize the surface of the gauze. This functionalized gauze has shown initial promise in mitigating cytotoxicity, especially under hypoxic conditions. The goal of this work is to investigate and demonstrate a proof of concept for a novel inorganic antioxidant functionalized gauze for reducing the oxidative stress in severe wounds, and ultimately, preserving tissue.
Materials Tests Articles and Analysis Methods
Images of the test materials are shown in Figure 2. Several polymer blends were tested to tune the release profile of the oxygen-producing hydrogel. This polymer may be embedded within the wound dressing or used to power an external pump to deliver oxygen through the dressing to the wound bed. The bioactive gauze is functionalized with an inorganic antioxidant material and is applied directly to the wound bed and serves to help stop bleeding like a typical gauze. However, the gauze is
 Figure 2. (A) Oxygen-producing polymer containing latent oxygen entrapped in a polymer matrix designed to release gaseous oxygen in a sustained manner over 72 h in response to water uptake from the wound exudate. (B) Bioactive gauze designed to be applied directly to the wound bed and scavenge for radical reactive oxygen species.
Figure 2. (A) Oxygen-producing polymer containing latent oxygen entrapped in a polymer matrix designed to release gaseous oxygen in a sustained manner over 72 h in response to water uptake from the wound exudate. (B) Bioactive gauze designed to be applied directly to the wound bed and scavenge for radical reactive oxygen species.also imbued with the ability to scavenge radical species that lead to further tissue death, thus preserving viable tissue while the soldier is transported to a hospital.
The release profiles from the oxygen-generating hydrogel were characterized using an oxygen displacement method. Briefly, oxygen release was activated by hydrating the polymer in a beaker. The oxygen generated was directed through tubinginto a graduated cylinder filled with water and fixed inverted in a larger beaker of water. The oxygen generated displaced the water and was measured by the cylinder graduations. Hydrogen peroxide decomposition was tracked as a measure of reactive oxygen scavenging capacity of the gauze. Several dilutions of the inorganic antioxidant were dispersed in hydrogen peroxide (5 mmol/ mL). As the inorganic antioxidant decomposed the hydrogen peroxide, the decrease in concentration was tracked by measuring UV absorbance at 240 nm. Likewise, the hydrogen peroxide decomposition capacity of the gauze was measured by immersing the approximately 100 mg pieces of gauze in 10 mL of 11 mM H2 O2 . The endpoint amount of H2 O2 decomposition was determined by measuring the amount of H2 O2 remaining after 10 min via absorbance at 240 nm.
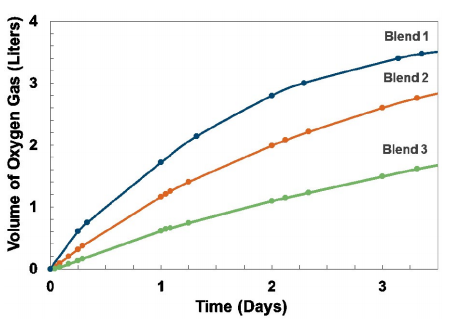 Figure 3. Oxygen release profiles measured via oxygen displacement for three polymer hydrogel blends.
Figure 3. Oxygen release profiles measured via oxygen displacement for three polymer hydrogel blends.Sustained Release of Oxygen Over 3 Days
The release profiles for three oxygen-producing polymer hydrogel blends show a near linear, tunable sustained release over 3 days (Fig. 3). By tuning the relative hydrophilicity of the polymer blend, various release profiles are achievable. Moreover, the material has a very high oxygen-producing capacity in a low-weight package. The polymer can produce over 3 L of gaseous oxygen over 3 days for every 100 g of active compound. These polymer hydrogels are currently being integrated into a conformal cover to yield an oxygen chamber that can deliver oxygen to otherwise hypoxic wounded tissue.
In Vitro Activity of the Inorganic Antioxidant Gauze
Hydrogen peroxide is an example of one ROS involved in oxidative stress in wounds. The active inorganic antioxidant showed dramatic ability to decompose hydrogen peroxide in vitro (Fig. 4). Doses as low as 3.2 mg/mL of the antioxidant could decompose 5 mmol/mL concentrations of hydrogen peroxide in about 10 min. Next, the same test was conducted on the functionalized gauze to see if the antioxidant maintained efficacy on
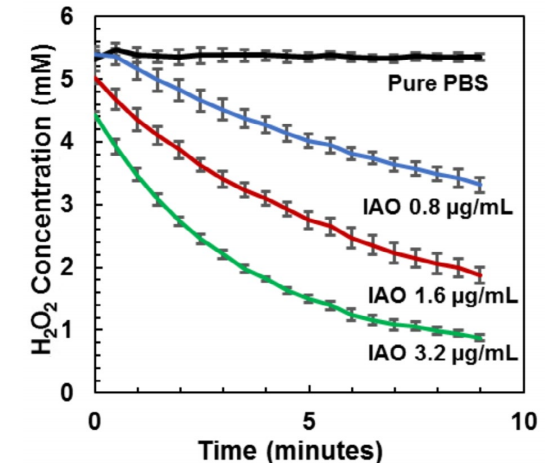 Figure 4. Kinetic hydrogen peroxide decomposition curves for various aqueous dilutions of inorganic antioxidant (IAO) mixed with 5 mM hydrogen peroxide. UV absorbance measurements at 240 nm were recorded in triplicate in a UV transparent 96-well plate every 30 s for 9 min.
Figure 4. Kinetic hydrogen peroxide decomposition curves for various aqueous dilutions of inorganic antioxidant (IAO) mixed with 5 mM hydrogen peroxide. UV absorbance measurements at 240 nm were recorded in triplicate in a UV transparent 96-well plate every 30 s for 9 min.the gauze. Figure 5 shows that a 100 mg piece of functionalized gauze decomposed 9.5 mmol/mL of the hydrogen peroxide in a 10 mL volume of 11 mM hydrogen peroxide within 14 min. This correlates roughly with the hydrogen peroxide decomposing capacity of about 70 units of catalase enzyme per gram of functionalized gauze. The unfunctionalized control gauze showed no change in hydrogen peroxide concentration.
Conclusions and Future
This work demonstrates how controlled release technology is being integrated into a lightweight, portable device that allow medics to supply oxygen to a wound and scavenge ROS while protecting the wound from the environment. Battelle will continue to integrate this technology with other bioactives to aid with fighting infection, preserving tissue, and arresting bleeding.
Acknowledgement
Funding for this work has been provided by the Office of Naval Research, Code 34 (Point of Contact, Dr. Timothy Bentley).
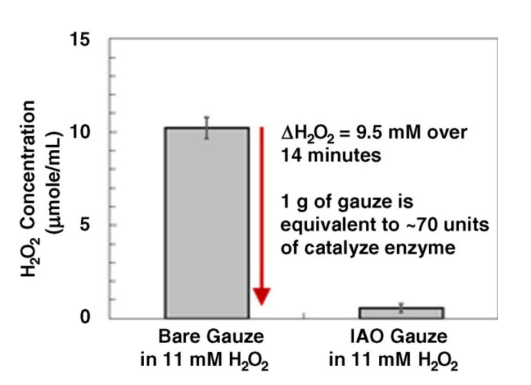 Figure 5. The endpoint hydrogen peroxide decomposition data for triplicate 100 mg samples of functionalized gauze immersed in 10 mL of 11 mM H2 O2 .
Figure 5. The endpoint hydrogen peroxide decomposition data for triplicate 100 mg samples of functionalized gauze immersed in 10 mL of 11 mM H2 O2 .References
1. Schreml, S, Szeimies, RM, Prantl, L, Karrer, S, Landthaler, M, Babilas, P. Oxygen in acute and chronic wound healing. Brit. J. Dermatol. 163: 257-268 (2010). 2. Oh, SH, Ward, CL, Atala, A, Yoo, JJ, Harrison, BS. Oxygen generating scaffolds for enhancing engineered tissue survival. Biomaterials 30: 757-762 (2009). 3. Dunnill, C, Patton, T, Brennan, J, Barrett, J, Dryden, M, Cooke, J, Georgopoulos, NT. Reactive oxygen species (ROS) and wound healing: The functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int. Wound J. 14: 89-96 (2017).


